 These include glass vials, filters, resin, tubing and disposable bags. *Complex multivalent vaccines can have production lead times of more than 36 months.ġ. Vaccine production can require more than 200 individual components, which are often manufactured in different countries. These strong quality requirements involve ad hoc pharmaceutical quality systems, quality assurance measures and procedures, several quality controls at each stage and an adequate infrastructure and separation of activities to guarantee vaccine identity, purity, sterility, efficacy and safety. Today, Pfizer manufactures more than 200 million. Successful manufacturing of high-quality vaccines requires international standardization of starting materials, production and quality control testing, and the setting of high expectations for regulatory oversight of the entire manufacturing process from start to finish, all while recognizing that this field is in constant change 1.Īll the components, manufacturing processes, testing methods, their reagents and standards have to comply with the standards defined for Good Manufacturing Practices (GMP). Pfizer is a proven, reliable multinational vaccine producer, supplying vaccines to more than 165 countries. 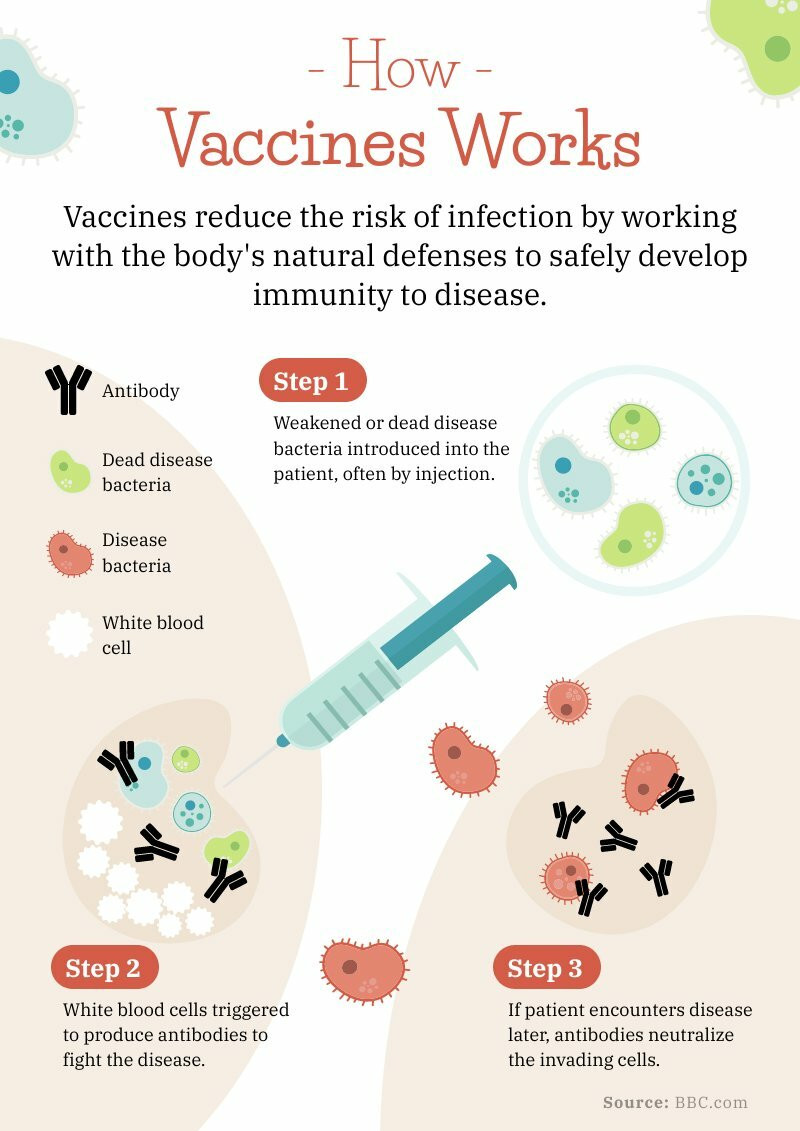
This allowed researchers from across the globe to analyze the virus and better understand how it causes disease. This sequence was quickly shared with other researchers. Earlier, Lonza did produce the vaccine for the EU, U.K. COVID-19 mRNA Vaccine Production Early in the COVID-19 pandemic, researchers used state-of-the-art genomic sequencers to quickly sequence the SARS-CoV-2 virus. The Vaccine Manufacturing Workshop for South-East Asia (SEAR) and Western Pacific Regions (WPR) ended today after two days of discussions centered on promoting quality and sustainable local production to improve access to vaccines in SEAR and WPR. (since the end of 2021) is in Geleen in the Netherlands, produced by its manufacturing partner Lonza. Once a vaccine has reached pre-approval stage following clinical trials, it is assessed by the relevant regulatory body for compliance with quality, safety and efficacy criteria. Another manufacturing site for the vaccines for the market outside the U.S. How a vaccine is approved for production For more information on the three phases of vaccine clinical trials, click here. The quality controls represent up to 70% of the full manufacturing duration. Besides CMOs, Moderna also manufactures the vaccine at its own production facility in Norwood, Massachusetts. Vaccines are complex biological products with lengthy manufacturing and control processes. The continued limited investment in these vaccines could be devastating for people’s lives. 
On average, it takes between 12-36 months* to manufacture a vaccine before it is ready for distribution. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
